 Our knowledge of the B vitamin folate has been undergoing major changes in the last ten years. We have come to the understanding that folic acid, a term often loosely identified as the same as folate, is in fact a synthetic oxidized form of folate.
Our knowledge of the B vitamin folate has been undergoing major changes in the last ten years. We have come to the understanding that folic acid, a term often loosely identified as the same as folate, is in fact a synthetic oxidized form of folate.
For many years (at least since the 1940s) the ONLY form of folate used in supplements and fortified foods has been folic acid. However, the good news for health is that the folate coenzymes of 5-MTHF and Folinic Acid are now readily available!
Recently, accumulated research has found that supplemental folic acid may actually accelerate cognitive decline in some older individuals. Folic acid also is being linked to increased risk of colon and rectal cancers, increased risk of childhood asthma born to folic-acid supplemented mothers, and accelerated growth of pre-existing cancers.
Unfortunately, journalists and even many medical professionals still haven’t figured out that folic acid is not the same as the naturally occurring vitamin folate. Even today, much of the medical mainstream, uses folic acid and folate as interchangeable terms. But folic acid is not the same as folate!
WHAT IS FOLATE?
Folate is a water-soluble B vitamin that is naturally present in foods. Folates are commonly consumed through a diet of green leafy vegetables, sprouts, fruits, brewer’s yeast, and animal products such as milk and dairy products, egg yolk and liver. Formerly known as folacin, folate is the generic term for both naturally occurring food folate and folic acid.
When consumed, food folates are often hydrolyzed to the monoglutamate form in the gut prior to absorption by active transport across the intestinal mucosa. Passive diffusion also occurs when pharmacological doses of folic acid are consumed. Before entering the bloodstream, the monoglutamate form is reduced to tetrahydrofolate (THF) and converted to either methyl or formyl forms. However, both of the metabolically active (coenzyme) forms 5-methyl tetrahydrofolate (5-MTHF) and 5-formyl tetrahydrofolate (also known as folinic acid) are found in foods and can enter the cells with no further modification.
Unfortunately folates contained in foods are unstable and susceptible to oxidation; they rapidly lose activity during food processing, manufacturing and storage and have a bioavailability range of 25–50%, depending on the kind of food. Fresh leafy vegetables stored at room temperature may lose up to 70% of their folate activity within three days and a cooking process in water can increase the loss to 95%.
Humans cannot synthesize folate and because of its water soluble nature, the body stores folate to a limited extent. For this reason folate represents a dietary requirement and must be consumed by diet.
FOLATE DEFICIENCY
Folate deficiency represents one of the most common nutritional deficiencies and may occur when dietary intake is inadequate, when an increased need is not matched by an increased intake (particular physiological conditions such as pregnancy, lactation, child growth), when there is altered absorption/excretion (or losses) and when metabolism or drug use interferes with the ability of the body to use folate.
Several conditions can lead to nutritional folate deficiency such as enzyme defects, malabsorption, digestive system pathology, liver disease but also conditions with a high rate of cell turnover such as rapid tissue growth (infants, kids and adolescents) pregnancy and lactation.
In severe cases deficiency can cause many clinical abnormalities, including macrocytic anemia, cardiovascular diseases, birth neural tube defects (NTDs) and carcinogenesis. Folate deficiency is associated with elevated levels of homocysteine, cerebrovascular and neurological diseases, and mood disorders.
MANY BENEFITS OF FOLATE COENZYMES
Folate coenzymes are responsible for the following important metabolic functions and benefits:
1) Formation of purines and pyrimidines, which in turn are needed for synthesis of the nucleic acids DNA and RNA. This is especially important during fetal development in the first trimester in preventing birth defects, such as neural tube defects.
2) Formation of heme, the iron-containing protein in hemoglobin
3) Interconversion of the 3-carbon amino acid serine from the 2-carbon amino acid glycine
4) Formation of the amino acids tyrosine from phenylalanine and glutamic acid from histidine
5) Formation of the amino acid methionine from homocysteine (Vitamin B12 as methylcobalamin also is needed for this conversion). Elevated levels of homocysteine have been implicated in a wide range of health disorders including atherosclerosis, osteoporosis, Alzheimer’s disease, and depression. In the reconversion of homocysteine to methionine the body uses the methionine to make the important amino acid s-adenosylmethionine (SAMe) which is known to be helpful in cases of depression.
6) Synthesis of choline from ethanolamine
7) Formation and maturation of red and white blood cells
8) Conversion of nicotinamide to N’-methylnicotinamide
Other conditions possibly benefiting from folinic acid supplementation include: AIDS/HIV, celiac disease, cervical displasia, cleft palate, Crohn’s disease, diarrhea, gout, high cholesterol, increased fracture of chromosomes, malabsorption and gastrointestinal inflammation, megaloblastic anemia, restless leg syndrome, postpartum depression, sprue, ulcerative colitis, and vitiligo.
Numerous drugs are known to inhibit the body’s ability to utilize folate, including: 1) aspirin, 2) cholesterol lowering drugs, 3) oral birth control pills, 4) antacids, and 5) methotrexate when used for rheumatoid arthritis. When taking these drugs (and many others) it is recommended that you take at least 800 mcg daily of folate, preferably as 5-MTHF and folinic acid. When taking folate it is recommended that you take adequate amounts of Vitamin B12 as methylcobalamin.
DISCUSSION OF FOLATE FORMS
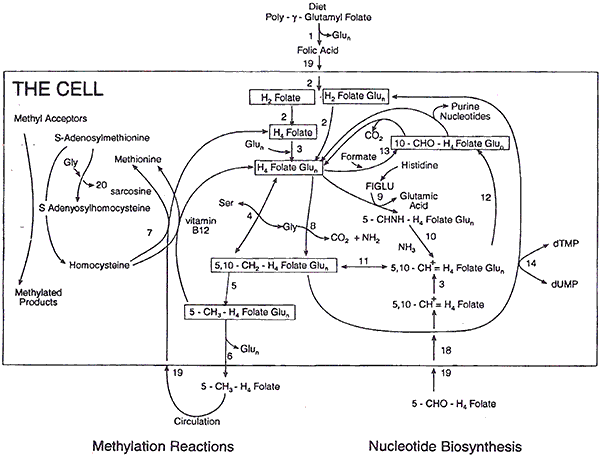
The figure shown below provides an overview of how the three forms of folate we will be discussing are metabolized in the cell. Basically the diagram shows that there are two major uses of folate in the body 1) Those dealing with methylation reactions and 2) those dealing with nucleotide biosynthesis, e.g., the production of DNA and RNA.
On the bottom left the diagram shows that 5-MTHF can directly enter the cell and be used for methylation reactions such as the conversion of homocysteine into methionine. On the bottom right the diagram shows that 5-formyl tetrahydrofolate (folinic acid) can directly enter the cell and be used for nucleotide biosynthesis after a few enzymatic conversions.
The top of the diagram shows that folic acid can enter the cell, but must go through a series of enzymatic conversions in order to accomplish what 5-MTHF and folinic acid can accomplish. The box in the lower middle of the diagram indicates where the MTHFR enzyme deficiency can block the metabolism of folic acid. The diagram shows that all of the important reactions can be accomplished by either 5-MTHF or folinic acid as they can be converted to one another by a series of enzymatic reactions. An important study (see abstract below) has indicated that the presence of a MTHFR enzyme deficiency does not impede the conversion of folinic acid into 5-MTHF.
FOLIC ACID: Once isolated and exposed to air natural folates in food becomes unstable and breaks down, and are generally no longer useful in nutrition. But a small amount of natural folate can be transformed by oxidation (a natural process) into folic acid, a much more stable form with a very long shelf life.
While human and animal cells cannot use the folic acid molecule itself in their normal metabolic processes, human cells (principally the liver) can transform folic acid back into many of its metabolically useful folate forms. That’s why folic acid—despite not being found in food—can do so much nutritional good, the best-known example being the prevention of birth defects including spina bifida, cleft lip, and cleft palate.
As we age, however, our bodies become increasingly slower at transforming folic acid into usefully metabolized folates. That’s probably the reason scientists are finding that folic acid (not folate) is associated with cognitive decline in the elderly. Some of these studies have shown significantly elevated levels of unmetabolized (and therefore not useful) folic acid building up in the bloodstreams of supplemented older individuals.
In addition to worsening folic acid metabolism with age, there are also a significant number (as high as 45 percent or more in some populations) of survivable human genetic defects of folate metabolism (MTHFR deficiency) which make it more difficult or, in some circumstances, impossible for sufferers to make metabolic use of folic acid.
So we believe that it is time to make folic acid supplements a part of history, and use only forms of naturally occurring folate when we use supplements. Although my company does produce a liquid folic acid supplement that has been especially useful for pregnant women who are experiencing gum problems, we have been making the coenzyme form Folinic Acid available for over 10 years and recently introduced the metabolically active form L-5-MTHF.
A small amount of folic acid (100–200 micrograms, the amount found in many multiple vitamins at present) is not likely to be a major problem for most people. However, more taken daily for years just might raise your long-term risk of colorectal cancer, cognitive decline, or other symptoms of elevated levels of homocysteine. If higher amounts are unavoidable (for example, until all prenatal vitamins switch from folic acid to folate), taking additional folate as 5-MTHF and Folinic Acid will very likely offset the folic acid still found in the multiple. In this regard, I have over the last five years eliminated folic acid from all of the multivitamins and B-Complex vitamins that we now make available.
FOLINIC ACID: also known as 5-formyl tetrahydrofolate, is one active form in a group of vitamins known as folates. In contrast to folic acid, a synthetic form of folate, folinic acid is one of the forms of folate found naturally in foods. In the body folinic acid may be converted into any of the other active forms of folate.
Compared to folic acid, folinic acid is expensive costing about 100 times more. However, the fact that the body only requires small amounts (less than one mg) means that one can obtain a two month supply of folinic acid for less than $10.
Folinic acid has been available as a supplement for more than 10 years and as such has been the form most used as a replacement for folic acid.
5-MTHF: also known as L-5-methyl tetrahydrofolate has been difficult to obtain until recently. An Italian company has made a patented form available (Quatrefolic®) that is combined with a vegetarian glucosamine. This form is particularly stable and highly bioavailable.
5-MTHF costs about 400 times more than folic acid. However, because the body requires less than one milligram (1 mg) on a daily basis, a person can buy a two-month supply for about $20.
5-MTHF is now readily available on the market, thereby making it possible to purchase at reasonable prices both coenzyme forms of folate. I have taken advantage of this in our Mighty Multi-Vite! multivitamin, which includes both 5-MTHF and folinic acid.
SUMMARY
Folic acid is not the same as folate. Folic acid presents problems in persons lacking the enzymes necessary to convert it into usable forms. Folic acid can also build up in the body in potentially toxic ways. Supplementing with naturally occurring coenzyme folates, such as 5-MTHF and folinic acid makes sense given advances in our understanding of how the body utilizes dietary folates.
ADDITIONAL RESOURCES
ABSTRACTS
J Nutr. 2000 Sep;130(9):2238-42.
Conversion of 5-formyltetrahydrofolic acid to 5-methyltetrahydrofolic acid is unimpaired in folate-adequate persons homozygous for the C677T mutation in the methylenetetrahydrofolate reductase gene.
Source
Vitamin Metabolism Laboratory, Jean Mayer U.S. Department of Agriculture Human Nutrition Research Center on Aging at Tufts University, Boston MA 02111, USA.
Abstract
Methylenetetrahydrofolate reductase (MTHFR) catalyzes the synthesis of 5-methyltetrahydrofolic acid (5-CH(3)-H(4) folic acid), the methyl donor for the formation of methionine from homocysteine. A common C677T transition in the MTHFR gene results in a variant with a lower specific activity and a greater sensitivity to heat than the normal enzyme, as measured in vitro. This study was undertaken to determine the capacity of homozygotes for the MTHFR C677T transition to convert 5-formyltetrahydrofolic acid (5-HCO-H(4) folic acid) to 5-CH(3)-H(4) folic acid, a process that requires the action of MTHFR. Six subjects homozygous for the C677T transition (T/T) and 6 subjects with wild-type MTHFR (C/C) were given a 5-mg oral dose of (6R:,S:)-5-HCO-H(4) folic acid. Plasma and urine were analyzed for 5-CH(3)-H(4) folic acid concentrations using affinity/HPLC coupled with fluorescence or UV detection. The mean areas under the curves created by the rise and fall of plasma 5-CH(3)-H(4) folic acid after the oral dose did not differ between the two genotypes, 424.5 +/- 140.3 (T/T) vs. 424.1+/- 202.4 h.nmol/L (C/C). There also was no significant difference in the mean cumulative 7-h urinary excretion of 5-CH(3)-H(4) folic acid between the T/T (2.5 +/- 1.4 micromol) and C/C (1.9 +/- 1.0 micromol) genotypes. Under the conditions employed, the conversion of oral 5-HCO-H(4) folic acid to 5-CH(3)-H(4) folic acid is not impaired in persons with the T/T MTHFR genotype. Possible reasons for these findings are discussed.
- PMID:
10958818
[PubMed – indexed for MEDLINE]


2 comments
Ever since I was diagnosed in 2013 with MTHFR heterozygous 677T, I have avoided supplemental folic acid, increased my consumption of cooked greens and experimented with both methylfolate and calcium folate (folinic acid) supplements (I also take sublingual mB12 daily). I found that even 400-800 mcg of daily methyfolate seems to be too much and eventually builds up and causes me to overmethylate (panic attack type symptoms — not pleasant!). Through trial and error, I have found supps that have calcium folinate (5-formyl-tetrahyrofolate [say that fast 5 times in a row, LOL]) give me the best results (300-600 mcg per day all but eliminate my RLS symptoms), but not being a chemistry student, I have had (and still have) trouble understanding the differences between methylated and non-methylated B9 supps.
This article is the best blend of technical info interspersed with plain English explanations of what the tech info means that I have found online. Thank you!